Among its many effects, adenosine binding to adenosine receptors in heart initiates a depressant effect. Adenosine receptor slows the heartbeat by slowing conduction in the atrioventricular node which is a region important to the pacing of the heart. Oxygen demand of heart tissues can be lowered by adenosine signaling to slow the heart. This is protective in times of stress, such as during periods of insufficient oxygenation of the heart. Due to this depressant effect on that heart tissue, adenosine is actually useful as a drug to treat certain arrhythmias that inappropriately activate the atrioventricular node.
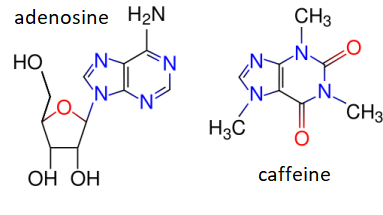
Adenosine receptors are also present in brain where they also have a depressant action. This can be reversed by caffeine, which due to its resemblance to the base portion of adenosine can bind in the adenosine binding pocket of the receptor and act as a competitive inhibitor. Blocking of a calming action is perceived by a person as stimulation, hence the apparent stimulant effect of caffeine. Due to its cardiac effects, the caffeine of coffee in excess may cause pounding of the heart.
Adenosine is the stick figure molecule shown below with its carbons in yellow, its nitrogens in blue, and its oxygens in red. It consists of a double ring base and a 5-carbon sugar. The adenosine is docked in an adenosine receptor whose structure had been determined through x-ray crystallography and is depicted as a ribbon diagram. In the figure, all amino acid side chains of the adenosine receptor that are within 4 Å of the adenosine molecule are also shown as stick figures. You will notice how nicely the adenosine fits into its binding site in the receptor.
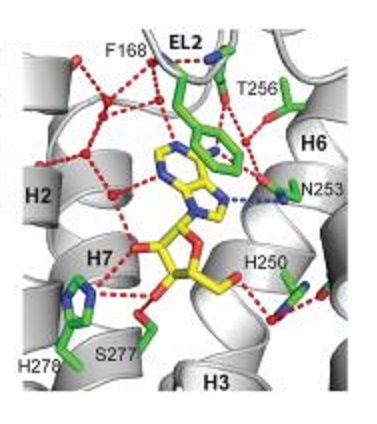
1. Near the top of the figure F168 is labeled with its side chain in green. This is a phenylalanine (amino acid single letter designation F) and is the 168th amino acid in the receptor counting from the amino terminus. Many bonds of the phenylalanine side chain ring are stacked closely atop the two rings of the base in the adenosine. The interaction between F168 and the adenosine is
2. Near the bottom of the figure serine S277 is labeled and colored in a similar manner with a red tip to denote its oxygen. Many carbons in the sugar of adenosine have hydroxyl groups (-OH) on them. The interaction between S277 and adenosine is
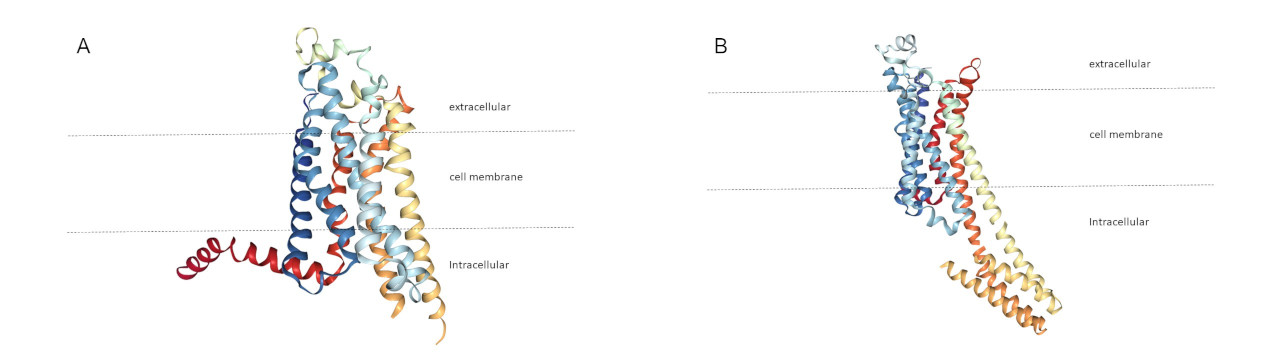
3. Below left is a ribbon drawing of a receptor in the absence of adenosine deduced by x-ray diffraction crystallography, and below right is the result of a similar experiment where adenosine is bound.

One of the below ribbon diagrams represents an adenosine receptor with an active adenosine analog bound, and the other represents an adenosine receptor with caffeine bound. Both are empty receptors so as not to give away an answer. Which of these best depicts the caffeine bound state?
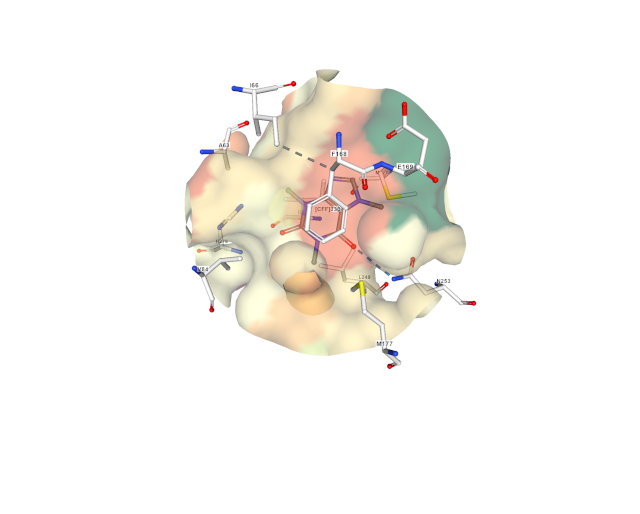
4. Below are two diagrams showing the normal ligand adenosine bound to the adenosine receptor. The multiple diagrams are to show the binding pocket from two different directions. The adenosine bonds are shown in gray with its carbons in blue and its oxygens in red. The adenosine receptor side chain bonds are shown in white. Four amino acid side chains of the adenosine receptor are shown, these are phenylalanine 168, glutamate 169, asparagine 253, and serine 277.
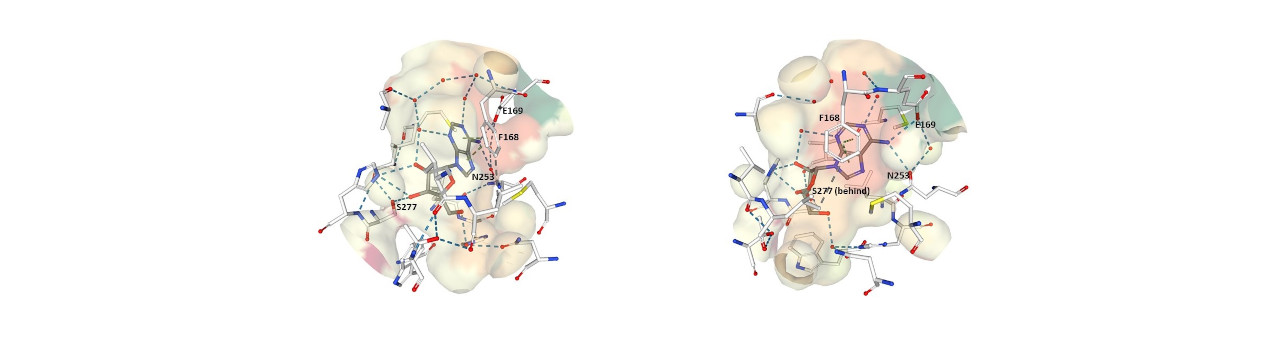
Which of these regions corresponds to a region of the receptor binding site that the ligand must interact with that causes the conformational shift in the protein that in turn brings about the intracellular response?
1. We are told in the problem that “Many bonds of the phenylalanine sidechain ring are stacked closely atop the two rings of the base in the adenosine.” We are told on page 8 of the required reading in Harper’s that van der Waals forces “act over very short distances, typically 2-4 Å.” These forces are sometimes called base stacking because they require a number of bonds in parallel with each other to reach significant strength. That is the situation here where the bonds in the ring of the phenylalanine are stacked over the bonds in the double ring base of adenosine.
2. We are told in the problem that “Many carbons in the sugar of adenosine have hydroxyl groups (-OH) on them.” We are told on page 6 of the required reading in Harper’s that “alcohols … can serve as both hydrogen acceptors and as donors of unshielded hydrogen atoms for formation of hydrogen bonds” Thus, the hydroxyl groups of the sugar are able to share hydrogens with the oxygen in the side chain of the nearby serine in the adenosine receptor.
3. This may be a trivial problem, but hopefully one still worth doing. Adenosine is known to propagate a signal across the cell membrane, and there is a visible change in the shape of the receptor protein (conformational shift) that is seen using x-ray crystallography comparing an adenosine receptor that is empty to an adenosine receptor occupied by an adenosine. Caffeine does not propagate the signal, so one would correctly expect that caffeine binding to receptor does not cause the conformational shift that adenosine does. Therefore, the caffeine bound protein shape is more like the free protein shape and less like the adenosine bound shape.
4. There are two key aspects here to realize. First, caffeine binds to the receptor without activating it (see figure next page). Second, caffeine binds to the receptor site that the similar adenosine double ring binds while it does not fill the receptor site that the sugar of the adenosine fills. This is a clue that where the sugar fits the receptor is important to bring about the conformational shift of the protein that triggers the intracellular response. One way of thinking about it is that adenosine sits in the driver’s seat and presses the brake pedal (depressant action). Caffeine sits in the driver’s seat, and may keep adenosine out of the driver’s seat, but caffeine does not press the brake pedal.
Phenylalanine 168 of the receptor engages with the double ring of adenosine or caffeine through van der Waals interactions, and asparagine 253 engages with the double rings through hydrogen bonding. Mutagenesis experiments have shown that glutamate 169 is essential for adenosine and caffeine to bind to the receptor. However, as explained above, it seems the interaction of the double ring is not sufficient to mediate the depressant action. In order to be activated, the receptor needs to interact with a bound signaling molecule at the location that the adenosine sugar molecule sits on the receptor. Receptor serine 277 is at that location and is in contact with the adenosine sugar due to hydrogen bonding. That interaction seems to participate in transducing the depressant action of adenosine. In the representation seen below of caffeine binding to adenosine receptor, serine 277 is not visible because it plays no role in caffeine binding.
Rodwell, V. W., Bender, D. A., Botham, K. M., Kenelly, P. J.. & Weil, P. A. (2018).
Image of 2YDO (Lebon, G., Warne, T., Edwards, P.C., Bennett, K., Langmead, C.J., Leslie, A.G.W., Tate, C.G. (2011) Agonist-Bound Adenosine A(2A) Receptor Structures Reveal Common Features of Gpcr Activation. Nature 474: 521) created with NGL (A.S. Rose, A.R. Bradley, Y. Valasatava, J.D. Duarte, A. Prlić, P.W. Rose (2018) NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 34: 3755–3758).
Image of 2YDV (Lebon, G., Warne, T., Edwards, P.C., Bennett, K., Langmead, C.J., Leslie, A.G.W., Tate, C.G. (2011) Agonist-Bound Adenosine A(2A) Receptor Structures Reveal Common Features of Gpcr Activation. Nature 474: 521) created with NGL (A.S. Rose, A.R. Bradley, Y. Valasatava, J.D. Duarte, A. Prlić, P.W. Rose (2018) NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 34: 3755–3758).
Image of 3RFM (Dore, A.S., Robertson, N., Errey, J.C., Ng, I., Hollenstein, K., Tehan, B., Hurrell, E., Bennett, K., Congreve, M., Magnani, F., Tate, C.G., Weir, M., Marshall, F.H. (2011) Structure of the adenosine A(2A) receptor in complex with ZM241385 and the xanthines XAC and caffeine. Structure 19: 1283-1293) created with NGL (A.S. Rose, A.R. Bradley, Y. Valasatava, J.D. Duarte, A. Prlić, P.W. Rose (2018) NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 34: 3755–3758).
Image of 5NM4 (Weinert, T., Olieric, N., Cheng, R., Brunle, S., James, D., Ozerov, D., Gashi, D., Vera, L., Marsh, M., Jaeger, K., Dworkowski, F., Panepucci, E., Basu, S., Skopintsev, P., Dore, A.S., Geng, T., Cooke, R.M., Liang, M., Prota, A.E., Panneels, V., Nogly, P., Ermler, U., Schertler, G., Hennig, M., Steinmetz, M.O., Wang, M., Standfuss, J. (2017) Serial millisecond crystallography for routine room-temperature structure determination at synchrotrons. Nat Commun 8: 542-542) created with NGL (A.S. Rose, A.R. Bradley, Y. Valasatava, J.D. Duarte, A. Prlić, P.W. Rose (2018) NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 34: 3755–3758).
Image of 5MZP (Cheng, R.K.Y., Segala, E., Robertson, N., Deflorian, F., Dore, A.S., Errey, J.C., Fiez-Vandal, C., Marshall, F.H., Cooke, R.M. (2017) Structures of Human A1 and A2A Adenosine Receptors with Xanthines Reveal Determinants of Selectivity. Structure 25: 1275-1285.e4) created with NGL (A.S. Rose, A.R. Bradley, Y. Valasatava, J.D. Duarte, A. Prlić, P.W. Rose (2018) NGL viewer: web-based molecular graphics for large complexes. Bioinformatics 34: 3755–3758).